Pressurized intrathoracic aerosol chemotherapy (PITAC): a new surgical technique for treatment of metastatic pleural effusion and pleural metastasis
Highlight box
Surgical highlights
• Pressurized intrathoracic aerosol chemotherapy (PITAC) is a new therapeutic option in patients with malignant pleural effusion (MPE) and/or pleural metastasis (PM). It’s performed in video-assisted thoracoscopy surgery through two ports. A CE-certified Reger NebulizerTM is inserted through the upper trocar and connected to an injector with chemotherapy solution. The system is left in steady-state for 30 min and after this period of nebulization, staff removes the remaining aerosol using a closed surgical smoke evacuation system.
What was conventional and what is novel/modified?
• In presence of MPE, possible therapeutic options include thoracentesis, indwelling pleural catheters and chemical pleurodesis.
• As a new alternative in the treatment of MPE and PM, PITAC, in addition to pleurodesis, associates an anti-neoplastic effect, by delivering into the chest of aerosol with chemotherapeutic agents.
What is the implication, and what should change now?
• PITAC is a safe, feasible and repeatable technique to control MPE recurrence and although promising in the field of therapy for PM, evaluated in the same way as patients having pressurized intraperitoneal aerosol chemotherapy, further investigations will be needed to evaluate the role in the prognosis of patients with MPE. Also, the promising outcomes of hyperthermic intrathoracic chemotherapy in patients with pleural recurrence of thymoma (combined with pleurectomy) and in patients with malignant pleural mesothelioma (combined with cytoreductive surgery), encourage new techniques as PITAC.
Introduction
Malignant pleural effusion (MPE) is an exudate characterized by the presence of malignant cells or tumor tissue and it is associated with a lower life expectancy (from 3 to 12 months after the initial diagnosis) (1).
Among the tumors that cause MPE, lung cancer is the most common cause, followed by breast cancer, malignant mesothelioma, ovarian neoplasm and lymphoma; yearly, there are 150,000 new cases in the United States and 100,000 in Europe (2).
MPE develops symptomatology characterized by dyspnea whose degree of severity is related to the quantity and speed of formation, cough and sometimes chest pain (3).
Physiologically, in the pleural cavity, there is no more than 10–20 mL of pleural fluid (PF); its accumulation occurs when invasion of the tumor involves the parietal pleura stomata and mediastinal lymph nodes so that absorption is reduced compared to production (4).
The diagnostic performance of cytology of the PF is close to 60%, with very slow sensitivity for mesothelioma (6%) and high for adenocarcinoma (79%); this because the tumor may involve the layer below the serous (1). Pleural biopsy so represents the gold standard procedure for diagnosis of MPE.
Possible therapeutic options include thoracentesis, indwelling pleural catheters and chemical pleurodesis (5). As a new alternative in the treatment of MPE and pleural metastasis (PM), pressurized intrathoracic aerosol chemotherapy (PITAC), in addition to pleurodesis, associates an anti-neoplastic effect, by delivering into the chest of aerosol with chemotherapeutic agents (6). We present this article in accordance with the SUPER reporting checklist (available at https://ccts.amegroups.com/article/view/10.21037/ccts-2025-1-54/rc).
Preoperative preparations and requirements
PITAC has been conceptualized in parallel with pressurized intraperitoneal aerosol chemotherapy (PIPAC), that in patients with peritoneal metastasis end-stage, induced objective histological regression preserving the quality of life and providing effective ascites control. Considering the results of PIPAC, the technique has also been applied to the pleura (6,7).
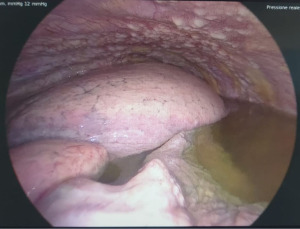
The procedure, as it is repeatable, can be applied in bilateral effusions but at different times to avoid the accumulation of chemotherapeutic agents. It is performed using VATS, allows direct administration of chemotherapy to pleural disease sites that would be difficult to reach with systemic administration and the intrapleural pressure induced by CO2 allows a better diffusion and a greater penetration of drugs inside the tumor cells with low systemic concentration (8) (Figure 1).
As pre-operative planning, computed tomography (CT) scan is required to valuate extension of MPE and possible presence of loculations.
PITAC is indicated for patients with an Eastern Cooperative Oncology Group (ECOG) performance status ≤2, without extra-thoracic metastases, age between 18 and 80 years, life expectancy of at least three months.
Occupational safety measures recommended PPE during PITAC procedures, include eye protection (plastic goggles or face shields), Filtering Facepiece (FFP) mask class 3 or N95 masks, reinforced surgical gown, double gloving with an inner pair resistant to chemotherapy and plastic overshoes. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki and its subsequent amendments. Written informed consent was obtained from all individuals included in this study, or their legal guardians or wards, for the publication of this article and the accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Step by step description
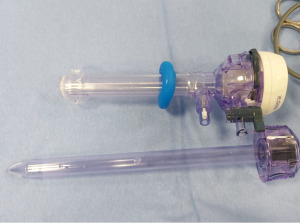
General anesthesia is required with monitoring of vital signs ( electrocardiogram, heart rate, blood pressure and oxygen saturation) and a double-lumen endotracheal tube is inserted to allow exclusion of the ipsilateral lung (correct positioning is checked under bronchoscopy); patient is positioned in the lateral decubitus position and two 12-mm balloon trocars (Figure 2) are inserted in the chest wall: one in the fifth intercostal space in the anterior axillary line (port of nebulizer), and the second one in the seventh intercostal space in the mid-axillary line (port of camera) (Figure 3).
Balloon trocars are required to create a closed system and to avoid dispersion of chemotherapy aerosol outside the thoracic cavity (Figure 4).
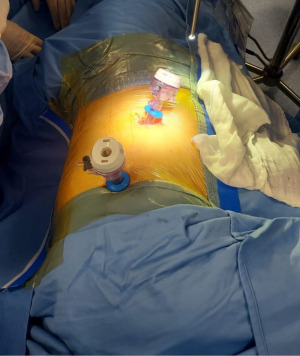
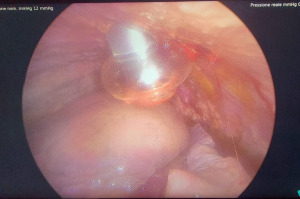
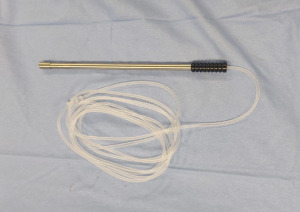
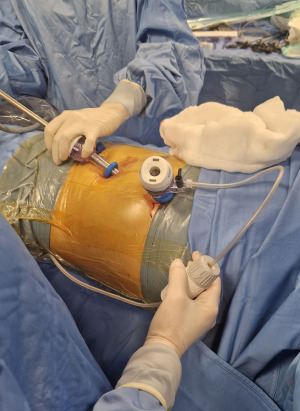
After suction of MPE, it is important to perform pleural biopsies for pathologic confirmation of pleural carcinomatosis and air-leak test prior to aerosolization; an intrathoracic pressure of 12 mmHg CO2 is maintained throughout the procedure to increase drug penetration into neoplastic tissue and a CE-certified Reger NebulizerTM (Figure 5) is inserted through the upper trocar and connected to an injector with chemotherapy solution (cisplatin 10.5 mg/m2 in 150 cc NaCl 0.9% and doxorubicin 2.1 mg/m2 in 50 cc NaCl 0.9%) according to the patient’s body surface area (BSA) calculated with the Boyd formula. These drugs are selected for cytotoxic and sclerosing effects on pleural layers. The patient is covered with a sterile drape and all staff leave operating room to avoid exposure to chemotherapy that is aerosolized into the pleural cavity using a remote-control at a flow of 0.7 mL/s with a maximal upstream pressure of 220 pounds per square inch (PSI) (Figure 6). The system is left in steady-state for 30 min with patient left alone with remote monitoring of vital signs; after this period of nebulization, staff enters in operating room to remove the remaining aerosol using a closed surgical smoke evacuation system with two microparticle filters to capture residual molecules (Figure 7). Balloon trocars are deflated and retracted and a 28-Fr chest tube is positioned in postero-apical position.
Postoperative considerations
Postoperative management is the same as for other VATS interventions. For safety of operators, drainage contained by chemotherapy, to the removal, needs for a dedicate waste chain for hazardous substances and material.
Results
We performed between May 2024 and January 2025, six PITAC for MPE of ovarian origin (n=1), lung origin (n=4) and breast origin (n=1). Informed consent was obtained for these cases. There were no complications during the procedures. No severe or life-threatening postoperative complications were reported; vomiting, chest pain and self-limiting subcutaneous emphysema subcutaneous were the only symptoms reported. PITAC was well tolerated (no signs of systemic toxicity) and all the patients had clinical benefits with no evidence of recurring MPE. Based on CT scans, patient 1 showed disease progression within 90 days and patients 3 and 4 within 30 days. Other patients showed stable disease for three months (Table 1).
Table 1
| Variables | Patient | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| Cancer origin | Ovary | Lung | Breast | Lung | Lung | Lung |
| Gender | Female | Male | Female | Female | Male | Female |
| Age (years) | 50 | 75 | 72 | 65 | 73 | 79 |
| ECOG PS | 2 | 1 | 1 | 1 | 1 | 0 |
| Histology | Serous adenocarcinoma | Squamous carcinoma | HER2+ ductal carcinoma | Adenocarcinoma | Squamous carcinoma | Adenocarcinoma |
| Other metastatic sites | No | No | No | No | No | No |
| Side | Left-sided | Right-sided | Left-sided | Left-sided | Right-sided | Left-sided |
| PITAC indication | MPE + PM (new diagnosis) | MPE + PM (new diagnosis) | MPE + PM (new diagnosis) | MPE + PM (new diagnosis) | MPE + PM (new diagnosis) | MPE + PM (new diagnosis) |
| Drained pleural effusion (cc) | 2,500 | 4,000 | 3,200 | 3,000 | 3,500 | 2,500 |
| Malignant cell in PE | No | Yes | No | No | Yes | No |
| Prior treatment | Systemic chemotherapy (three lines) | No | Immunotherapy | No | No | No |
| Prior pleurodesis | No | No | No | No | No | No |
| Other treatments after PITAC | Chemotherapy | Chemotherapy | Immunotherapy | Chemotherapy | Chemotherapy | Immunotherapy |
| Concurrent surgeries | PIPAC | None | None | None | None | None |
| Intraoperative complications | None | None | None | None | None | None |
| Postoperative complication | None | None | None | None | None | None |
| Postoperative chest pain | 2 | 3 | 1 | 2 | 2 | 1 |
| Systemic absorption of drug | No | No | No | No | No | No |
| Creatinine levels | Normal | Normal | Normal | Normal | Normal | Normal |
| Discharge after PITAC (days) | 6 | 3 | 7 | 6 | 3 | 7 |
| Recurring MPE (CT scan) | No at 90 days | No at 45 days | No at 30 days | No at 30 days | No at 60 days | No at 90 days |
CT, computed tomography; ECOG PS, Eastern Cooperative Oncology Group performance status; MPE, malignant pleural effusion; PE, pleural effusion; PIPAC, pressurized intraperitoneal aerosol chemotherapy; PITAC, pressurized intrathoracic aerosol chemotherapy; PM, pleural metastasis.
Discussion
In this article, we describe as PITAC is a safe, feasible and repeatable technique to control MPE recurrence and although promising in the field of therapy for PM, evaluated in the same way as patients having PIPAC, further investigations will be needed to evaluate the role in the prognosis of patients with MPE. Also, the promising outcomes of hyperthermic intrathoracic chemotherapy (HITOC) in patients with pleural recurrence of thymoma (combined with pleurectomy) and in patients with malignant pleural mesothelioma (combined with cytoreductive surgery), encourage new techniques as PITAC (8).
There are few data in literature; however, a Danish study (Hansen et al.) on a patient who underwent three PITAC for MPE from HER2+ ductal breast cancer showed the absence of malignant cells in the PF after the third procedure, while before PITAC1 (first PITAC), malignant cells were detectable. In analogy with Peritoneal Regression Grading Score (PRGS), a four-tiered Thoracic Regression Grading Score (TRGS) was assessed for pleural biopsies: TRGS-1 equals complete histological response, and is defined as the absence of residual cancer and harbours only regressive histological features, TRGS-2 is defined as a biopsy where regressive changes are predominant over cancer cells (major response), TRGS-3 is a biopsy where cancer cells are predominant but regressive changes are present (minor response), and TRGS-4 is a biopsy with cancer but without histological features of regression (no response). In a patient with rectum adenocarcinoma, in a study by the same authors, pleural biopsies demonstrated regression from TRGS 3 prior to PITAC1 to TRGS 1 prior to PITAC2 (second PITAC). These cytological and histological data suggest that PITAC may induce regression of pleural disease, but at present no other data are available (9,10).
If PITAC in patients with MPE can be an alternative treatment to repeated thoracentesis, indwelling pleural catheters and talcum pleurodesis, the data we have today in literature don’t allow us to say this, however in our experience we don’t detect intraoperative complications, evidence of recurring MPE and 30-day mortality was zero. Complications reported in literature are bleeding (for the pleural biopsy) and air-leak. No severe or life-threatening post operative complications were reported.
Conclusions
PITAC is a safe, feasible and repeatable technique to control MPE recurrence and that seems to induce, in some patients, histological and cytological response. However, further studies will be needed to confirm the benefit in improving the prognosis of patients with MPE and/or PM.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the SUPER reporting checklist. Available at https://ccts.amegroups.com/article/view/10.21037/ccts-2025-1-54/rc
Peer Review File: Available at https://ccts.amegroups.com/article/view/10.21037/ccts-2025-1-54/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://ccts.amegroups.com/article/view/10.21037/ccts-2025-1-54/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki and its subsequent amendments. Written informed consent was obtained from all individuals included in this study, or their legal guardians or wards, for the publication of this article and the accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Ferreiro L, Suárez-Antelo J, Álvarez-Dobaño JM, et al. Malignant Pleural Effusion: Diagnosis and Management. Can Respir J 2020;2020:2950751. [Crossref] [PubMed]
- Gonnelli F, Hassan W, Bonifazi M, et al. Malignant pleural effusion: current understanding and therapeutic approach. Respir Res 2024;25:47. [Crossref] [PubMed]
- Skok K, Hladnik G, Grm A, et al. Malignant Pleural Effusion and Its Current Management: A Review. Medicina (Kaunas) 2019;55:490. [Crossref] [PubMed]
- Barone M, De Leo G, Barresi P, et al. Ascite e versamenti pleurici recidivanti. Available online: https://hdl.handle.net/11570/1581587
- Hansen PS, Graversen M, Detlefsen S, et al. Review on treatment of pleural metastasis and malignant pleural effusion with Pressurized IntraThoracic Aerosol Chemotherapy (PITAC). Pleura Peritoneum 2024;9:47-53. [Crossref] [PubMed]
- Drevet G, Maury JM, Bakrin N, et al. Technique of pressurized intrathoracic aerosol chemotherapy (PITAC) for malignant pleural effusion. Pleura Peritoneum 2020;5:20200129. [Crossref] [PubMed]
- Di Giorgio A, Macrì A, Ferracci F, et al. 10 Years of Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC): A Systematic Review and Meta-Analysis. Cancers (Basel) 2023;15:1125. [Crossref] [PubMed]
- Hofmann HS, Ried M. Hyperthermic intrathoracic chemotherapy in thoracic surgery. Chirurg 2019;90:681-94. [Crossref] [PubMed]
- Khosrawipour V, Mikolajczyk A, Paslawski R, et al. Intrathoracic aerosol chemotherapy via spray-catheter. Mol Clin Oncol 2020;12:350-4. [Crossref] [PubMed]
- Hansen PS, Graversen M, Detlefsen S, et al. Pressurized IntraThoracic Aerosol Chemotherapy (PITAC) directed therapy of patients with malignant pleural effusion and pleural metastasis. Pleura Peritoneum 2024;9:131-9. [Crossref] [PubMed]
Cite this article as: Familiari D, Monaco F, Milone E, Rizzo R, Valenti E, Nunnari FA, Marando R, Macrì F, Musumeci G, Macrì A. Pressurized intrathoracic aerosol chemotherapy (PITAC): a new surgical technique for treatment of metastatic pleural effusion and pleural metastasis. Curr Chall Thorac Surg 2026;8:2.